Polyolefins
Polyolefins is a major class of synthetic polymers made by the polymerization of an olefin (alkene) or its suitable derivative. Most of these are obtained from petro chemical industry. Polyethylene, polypropylene, PVC, Teflon, etc. belong to the class of polyolefins.
Polyethylene or Polyethene
It is formed by polymerization of ethylene (CH2=CH2). Polyethene is of two types - Low Density Polyethene (LDPE) and High Density Polyethene (HDPE) depending upon the nature of has branching in polymer chain and is not compact in polymer molecules. Low density polyethene has branching in polymer chains and is not compact in packing. High density polyethere has linear chain of molecules which are packed in a more compact fashion. Polyethylene is used for making pipes, insulators, packing films, carry-bags, etc.
Polypropylene
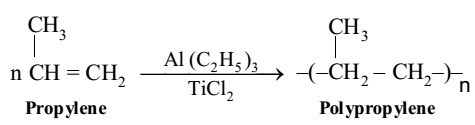
The monomer units are propylene molecules. It is generally manufactured by passing propylene through n-hexane (inert solvent) containing Ziegler Natta catalyst (a mixture of triethyl aluminium and titanium chloride). Polypropylene is harder, stronger and lighter than polyethene. Polypropylene is used for packing of textile material and food, lining of bags, gramophone records, ropes, carpet fibres, etc.
Teflon or Polytetrafluoro ethylene (PTFE)
The monomer unit is terafluoroethylene molecule. Teflon is prepared by heating tetra fluoroethylene under pressure in the presence of ammonium peroxosulphate. Teflon is a very tough material and is resistant towards heat, action of acids or bases. It is bad conductor of electricity. Teflon is used for coating utensils to make them non-sticking, making seals and gaskets which can with stand high pressures, insulation for high frequency electrical installations.
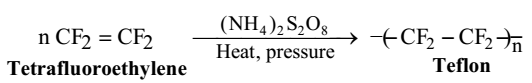
Polyvinylchloride (PVC)
The monomer units are vinyl chloride molecules. PVC is prepared by heating vinyl chloride in an inert solvent in the presence of dibenzoyl peroxide. PVC is a hard horny material. However, it can be made to acquire any degree of pliability by the addition of a plasticizer. It is resistant to chemicals as well as heat. It is used for making rain coats, hand bags, toys, hosepipes, gramophone records, electrical insulation and floor covering.

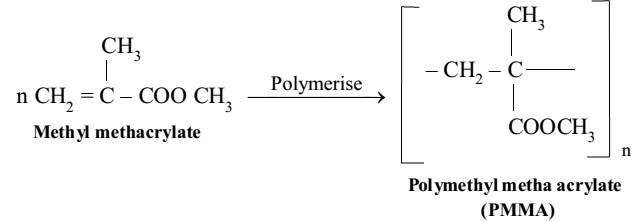
Polymethyl Methacrylate (PMMA)
Its monomer unit is methyl methacrylate. PMMA is a hard and transparent polymer and quite resistant to the effect of heat, light and ageing. It has high optical clarity. It is used in the manufacture of lenses, transparent domes and skylights, dentures, aircraft windows and protective coatings. Its commercial names are Lucite, Plexiglass, Acrylite and Perspex.