Effects of Heat
When a body is heated changes may occur in some of its properties .These changes are the effects of heat.
Rise in temperature
When a body is heated its temperature increases, that is why, it appears warmer when touched.
Change of state
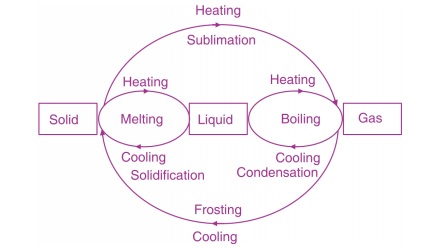
When heat is supplied to a substance in solid state, its temperature rises till at a particular temperature it may change into its liquid state without any further change in its temperature. This characteristic constant temperature at which a solid changes into its liquid state is called melting point of the solid. The melting point of a substance is a characteristic, constant value and different substances may have different values of melting points.
Conversion of a solid into its liquid state at its melting point is called change of state from solid to liquid (fusion) and the heat that is transferred to the substance during melting is called Latent Heat of Fusion. It does not becomes apparent in the form of rise in temperature. Latent heat of fusion of a solid substance is defined as the amount of heat (in joules) required to convert 1 kg of the substance from solid to liquid state at its melting point.
Similarly, when heat is supplied to a substance in liquid state its temperature rises but there is a possibility that it changes into its vapour state at a constant temperature. The heat supplied in this case is called Latent Heat of Vaporization. Latent heat of vaporization of a liquid is defined as the amount of heat (in joules) required to convert 1 kg of the substance from its liquid to gaseous state at a constant temperature. Latent heats of vaporization of different substances are also different.
Vaporization may take place in two different ways:
- Evaporation from the surface of a liquid at any temperature
- Boiling of the whole mass of the liquid at a constant temperature called boiling point of the liquid. Boiling points of different liquids are also different.