Matter
Matter is any thing which has mass and occupies space. All solids, liquids and gases around us are made of matter. Atoms is a basic unit of matter and all chemical properties of matter can be explained on its basis.
States of Matter
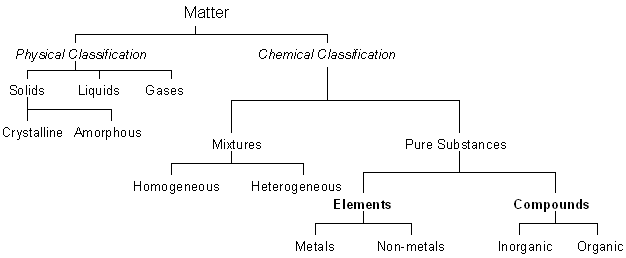
Matter can be classified in many ways:
- by the physical state of matter as a solid, liquid, or gas
- by the chemical composition of matter as an element, compound or mixture
Matter can ordinarily exist in three states - solid, liquid and gas. These three states of matter have different properties. Water exists in all the three states namely steam or water vapour (gas), water at room temperature (liquid) and ice (solid). This is the only substance which exists naturally in all the three states.
The characteristic properties of different states of matter depend on intermolecular forces. The forces holding molecules together are called intermolecular forces. Intermolecular forces (forces between the constituent molecules) try to keep molecules together but thermal energy always tries to keep them far apart. It is the competition between molecular interaction energy and thermal energy that decides whether a given substance under given conditions will be a solid, liquid or gas.
Thermal or heat energy can convert one state of matter into another state. Thus, a particular state of a matter depends on both intermolecular force and the thermal energy which basically depends upon temperature.
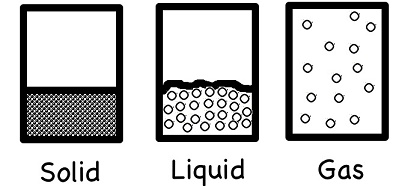
Solids
A solid has definite size and shape which do not change on their own. However, by using external forces you can change the shape of a solid.
In solids, the constituent particles are present very close to each other and the intermolecular forces operating between the constituent particles are very strong and they are capable of keeping the molecules in fixed positions. This is the reason why solids are rigid and hard.
When a solid is heated there is an increase in thermal energy of the particles which results in conversion of solid into liquid. The temperature at which this happens is the melting point of the solid.
Liquids
A liquid has a definite volume. However, a liquid does not have a definite shape. It takes the shape of its container.
Liquids have properties intermediate between solids and gases. The intermolecular forces in liquids are weaker than solids but stronger than gases. In liquids the constituent particles do not occupy fixed position as in solids, but they have freedom of movement as in gases. In liquids intermolecular forces are stronger than those of gases.The constituent particles (atoms and molecules) in a liquid can break away from each other and get attracted while approaching the other molecules.
Gases
A gas occupies the entire volume of the container irrespective of its size. In gases, molecules move freely because the intermolecular forces are very weak and are unable to keep the gas molecules together in bulk. The molecules remain far apart from each other due to weak molecular interactions. Since molecules are far away from each other in gases, they can be brought closer when pressure is applied. This is the reason why-gases are highly compressible.
When temperature increases, volume of the gas also increases. For example when a closed container is heated it blasts due to rapid increase in volume.
Characteristics of States of Matter
| Solid | Liquid | Gas | |
| Volume | Fixed | Fixed | No Fixed |
| Density | High | Moderate | Low |
| Shape | Definite Shape | Shape of Container | No Definite Shape |
| Fluidity | Does not Flow | Flows Smoothly | Flows Smoothly |
| Cmpressibility | Negligible | Small | High |
Effect of Temperature and Pressure
When heat is supplied to a solid, it expands. After receiving thermal energy, particles (atom/molecules) vibrate more rapidly in their position and take up more space. If particles become more energetic on further heating they leave their fixed positions and the solid melts.
When a liquid is heated, it is converted into a gas. This happens because the kinetic energy of the particles becomes so high that they can overcome the intermolecular force within the liquid.

When a gas is heated, kinetic energy of the particles increases. They move more freely and at much higher speed. Intermolecular distance also increases and the volume of the gas increases if pressure is kept constant.
Conversion of pure substance from solid to liquid takes place at one particular temperature. This particular temperature is called melting point of that particular solid substance.
Similarly when the liquid cools down, it converts into solid at a particular temperature. This temperature is called freezing point of that particular liquid substance. The temperature at which a liquid boils and is converted into a gas is boiling point of the liquid.
Elements, Compounds and Mixtures
Elements
All substances are made up of chemical elements. A chemical element is a basic form of matter that cannot be chemically broken down into simpler substances. A chemical element is a pure substance and it consists of one type of atom distinguished by its atomic number. Examples of some elements are : helium, carbon, iron, gold, silver, copper, aluminum, hydrogen, oxygen, nitrogen, sulphur, copper, chlorine, iodine, uranium, and plutonium.
Compounds
A compound is a substance formed when two or more than two elements are chemically combined. A compound can be defined as a pure substance made from two or more elements chemically combined together in a definite proportion by mass. When elements join to form compounds they lose their individual properties. Compounds have different properties from the elements they are made of. For example, water (a compound) is made up of elements - hydrogen and oxygen but properties of water are different from those of hydrogen and oxygen.
Mixtures
Mixtures are broadly divided in two major groups:
- Homogeneous mixtures
- Heterogeneous mixtures
Homogeneous Mixture
In some mixtures, constituents are completely mixed in such a way that the entire mixture has the same composition throughout. Such mixtures which have uniform composition are called homogeneous mixtures. For example, by mixing sugar and water, the entire mixture has the uniform sweetness. Technically such homogeneous mixtures are called solutions. For example common salt, which is solid when dissolved in water, forms a liquid mixture or a salt solution.
A homogeneous mixture is a mixture where the substances are completely mixed together and have uniform composition throughout.
Types of Homogeneous Mixtures
Solid + Liquid: Solid dissolves in liquid to form transparent solution. For example, sugar in water or salt in water, iodine in ethyl alcohol (tincture iodine)
Liquid + Liquid: It forms a single transparent mixture. For example, mixture of water and ethyl alcohol.
Gas + Liquid: Gas completely dissolves in a liquid to form a transparent solution. For example, soda water and any other common soft drink.
Gas + Gas: Mixture of two or more gases. For example, Air.
Solid + Solid: Some metallic alloys like brass, bronze.
Heterogeneous Mixture
Mixtures where the constituents do not completely mix with each other, and remain separate, are called heterogeneous mixtures. In such mixtures, one substance is spread throughout the other in the form of small particles, droplets or bubbles.
A heterogeneous mixture is a mixture where the substances (parts or phases) remain separate and composition is not uniform.
Types of Heterogeneous Mixtures
Suspension: Solid + Liquid. For example, flour in water, river water carrying mud
Gel: Liquid trapped in solid. For example, fruit jelly, agar gel
Emulsion: Mixture of tiny droplets of one liquid suspended in another. For example, milk
Aerosol: Small droplets of liquid or particles of solid dispersed in a gas. For example, clouds (liquid in gas) and smoke (solid in gas)
Foam: Gas in liquid - small bubbles of gas trapped in liquid. For example, shaving foam
Foam: Gas in solid - small bubbles of gas trapped in solid. For example, polystyrene foam (Thermocoal)
Separation of Mixtures
To separate different components of a mixture variety of physical techniques are available. All these separation techniques are based on difference in the physical properties of the components present in the mixture.
Separation by using Separating Funnels
The mixture of two immiscible liquids (i.e, the liquids that do not mix, as oil and water) can be separated by using a separating funnel. The mixture is placed in separating funnel and allowed to stand for some times. When the two layers of liquids are separated, the denser liquid which is in the lower part, is first collected by opening the stop-cock. This method is very useful in industries.
Separation by Evaporation
The separation of liquid (solvent) and solid (solute) from a solution is done by removing the liquid (solvent) by heating or by solar evaporation. By evaporation you can recover the solute component only in solid or powder form.
Separation by Filtration
Filtration is a better method for separating solids from liquids in heterogeneous mixtures. In filtration the solid material is collected as a residue on filter paper and the liquid phase is obtained as filtrate. The method of filtration is used on a large scale in industries.
Separation by Crystallization
Crystallization is a process of formation of solid crystals from a solution. The method of crystallization for separating solid from liquid begins by evaporating the liquid. However, in crystallization, the evaporation is stopped when the solution is concentrated enough. The concentrated solution thus produced, is allowed to cool slowly to form crystals which can be separated by filtration.
Separation by Distillation
The method of distillation is used to separate a liquid from a solution of a homogeneous mixture. The distillation is a process in which a liquid or mixture of liquids is boiled in a distillation flask. The vapour is condensed by passing through a water-cooled tube called condenser and collected as liquid called distillate.
In case of a solution of two miscible liquids (the liquids which can be mixed completely) the separation is based on the fact that the liquids will have different boiling points and there is a wide difference between the boiling points of the two liquids.
Separation based on Magnetic Properties
In a mixture of magnetic and non-magnetic substances, the magnetic substance can be separated by using a magnet. In industry, this method is used to separate iron materials from non-magnetic materials by using large electromagnets.