Source of Metals and Metallurgy
The earth crust is the major source of metals. Some metal salts are also present in sea. The constituents of earth crust which contain these metals or their compounds are known as minerals.
At some places minerals contain a high percentage of a particular metals and the metal can be profitably extracted from it, such minerals are called ores. An ore taken out from the earth contains a lot of impurities in form of sand and other undesirable materials. Metal is present in these ores in form of a compound.
Getting pure compound of a metal from its ore and finally getting the metal from it pure compound is called metallurgy.
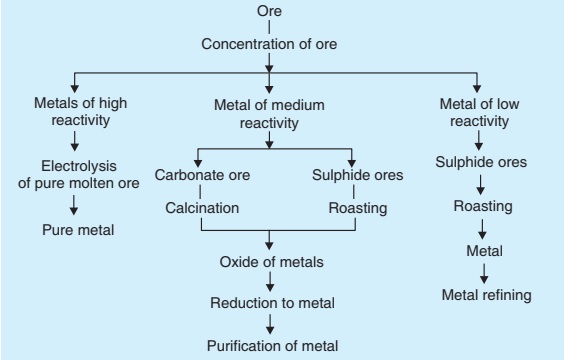
Metals are broadly considered in three categories:
- lower part of activity series (metal of low reactivity)
- middle part of activity series (metals of medium reactivity)
- top part of activity series (metal of high reactivity)
Metals in the lower part of the activity series are very unreactive. For example, mercury which is obtained as HgS (cinnabar) can be extracted easily.
2HgS + 3O2 (g) + heat → 2HgO (s) + 2SO2 (g)
On further heating, HgO is decomposed in mercury and oxygen.
2HgO (s) + heat → 2Hg (l) + O2 (g)
Metals in the bottom of activity series like Ag, Au are least reactive and are found in native state.
The metals in the middle of the activity series such as iron, zinc lead are moderately reactive. They are present usually as sulphide or carbonate in nature. Prior to reduction, these ores are converted into oxides as it is easy to reduce metal oxides.
Roasting: 2ZnS (s) + 3O2 (g) + heat → 2ZnO (g) + 2SO2 (g)
Calcination: ZnCO3 (s) + heat → ZnO (s) + CO2 (g)
Metal oxides are reduced to corresponding metal using carbon.
ZnO (s) + C (s) → Zn (s) + CO (g)
Obtaining metal from their compounds in always a reduction process.
We also use displacement reaction for reduction of metal oxide. For example,
Fe2O3 (s) + 2Al (s) → 2Fe (l) + Al2O3 (s)
This type of reaction is also known as thermite process and is very useful in welding of rail tracks or other heavy machineries.
Metal at the top of activity series are highly reactive. These metals have high affinity for oxygen and therefore can not be obtained by reduction with carbon. These metals (such as Na, K, Mg) are obtained by the process of electrolysis of their molten salt.
Even Al is also obtained by electrolysis of its oxide (Al2O3). Sodium is obtained by electrolysis of its molten salt, NaCl.
At the cathode: Na+ (l) + e- → Na (s)
At the anode: 2Cl- (l) ⎯⎯→ Cl2 (g) + 2e-