Surface Energy
The surface layer of a liquid in a container exhibits a property different from the rest of the liquid. A molecule, say P, well inside the liquid is attracted by other molecules from all sides. However, it is not the case for the molecules at the surface.
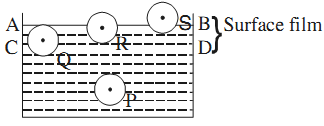
Molecules S and R, which lie on the surface layer, experience a net resultant force downward because the number of molecules in the upper half of sphere of influence attracting these molecules is less than those in the lower half.
If any liquid molecule is brought to the surface layer, work has to be done against the net inward force, which increases their potential energy. This means that surface layer possesses an additional energy, which is termed as surface energy.
For a system to be in equilibrium, its potential energy must be minimum. Therefore, the area of surface must be minimum. That is why free surface of a liquid at rest tends to attain minimum surface area. This produces a tension in the surface, called surface tension.
Surface tension is a property of the liquid surface due to which it has the tendency to decrease its surface area. As a result, the surface of a liquid acts like a stretched membrane.
SI unit of surface tension is Nm–1 and its dimensions are [MT–2].