Acidic & Basic Nature of Phenols
Acidic Nature
Phenols are much more acidic than alcohols. Since phenols are acidic in nature, they are soluble in dilute sodium hydroxide.

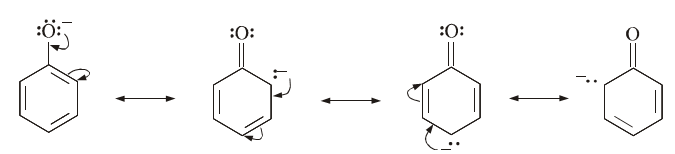
Resonance of Phenoxide Ion
The greater acidity of phenols can be attributed to the resonance stablisation of the phenoxide ion. The delocalisation of the negative charge over the benzene ring stabilises the phenoxide ion. No such stabilisation is possible, in case of alkoxide ions.
Basic Nature
Phenols behave as weak bases also. Similar to alcohols, they can also be protonated to give phenyloxonium ion.
