Acidity of Carboxylic Acids
Carboxylic acids are acidic in nature. They dissociate in water according to following equilibrium to give a proton and the carboxylate ion.
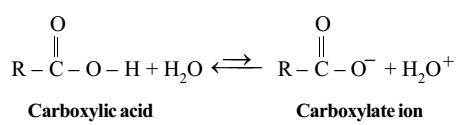
Carboxylic acids are much more acidic than alcohols. The carboxylate ion obtained by the dissociation of carboxylic acids can be represented as a resonance hybrid.

These structures show that the negative charge is delocalized over two oxygen atoms. Thus, the carboxylate ion gets stabilized. The greater stability of carboxylate ion facilitates
the release of proton from the –COOH group. No such resonance stabilization is possible in the alkoxide ion.
The electron-donating substituents decrease the acidity of carboxylic acids. Thus, ethanoic acid is less acidic than methanoic acid. The alkyl groups are electron releasing in nature, they make the release of H+ difficult and hence decrease the acidity.