Carbon Atom in Organic Chemistry
Electrons are found in regions around the nucleus in an atom. and those regions are called orbitals. The orbitals can be defined and differentiated by size, shape, and orientation. Valence electrons are electrons that are found in the outermost shell. The carbon atom has four valence electrons. These valence electrons are involved in chemical reactions and bonding.
Electronic Configuration
Atomic Number: 6
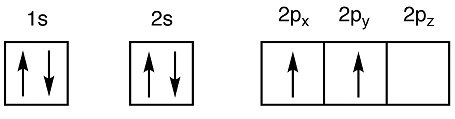
The six electrons of a carbon atom are distributed in the orbitals as:
Electronic Configuration: 1s22s22p2
Ground State of Carbon Atom
Hybridization of Orbitals
sp3 Hybridization in Methane
The carbon atoms of alkanes are sp3 hybridized. In order to form the four bonds in methane, a carbon atom needs four half-filled orbitals. In order to have more free half-filled orbitals, the carbon atoms undergo hybridization. Since the latter has higher energy than the former, this change requires energy.

In the excited-state carbon atom, there are four valence orbitals each bearing one electron (2s, 2px, 2py, and 2pz). This excited-state of carbon atom can form covalent bonds with four hydrogen atoms, resulting in a methane molecule. The four sp3 hybrids are directed to the corners of a tetrahedron with bond angles of 109.5°.
sp2 Hybridization in Ethene
The coordination number of each carbon atom in the ethylene molecule is three, not four as is the case with the carbon atoms in methane. In carbon-carbon double bonds, the carbons undergo sp2 hybridization. In this hybridization, only one 2s, and two 2p orbitals are involved.

The C=C contains a sigma (σ) bond and a pi (π) bond. The pi bond is formed by the unhybridized 2p orbital overlap. The three equal hybrids lie in an xy-plane with bond angles of 120°.
sp Hybridization in Ethyne
An sp hybridized carbon atom is bonded only to two other atoms. In this type of hybridization, one 2s orbital and one 2p orbital are involved. A carbon-carbon triple bond contains one sigma bond and two pi bonds.
