Conversion of Alcohols to Alkyl Halides
Alcohols are converted into haloalkanes by treating with
- (a) hydrogen halides
- (b) phosphorus halides
- (c) thionyl chloride
Reaction with Hydrogen Halides
Hydrogen halides react with an alcohol in presence of a dehydrating agent such as anhydrous zinc chloride to produce a haloalkane.
Chloroethane is prepared by the reaction of ethanol with concentrated hydrochloric acid in presence of anhydrous zinc chloride. Zinc chloride absorbs water from the reaction mixture and thus prevents the reverse reaction.
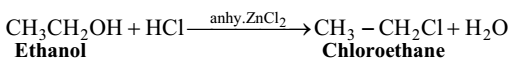
Reaction with Phosphorus Halides
Haloalkanes are conveniently prepared by the reaction of an alcohol with a phosphorus halide (PCl3, PCl5, or PBr3).

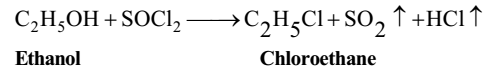
Reaction with Thionyl Cholride
Thionyl chloride (SOCl2) reacts with an alcohol to yield a chloroalkane. As both the byproducts, SO2 and HC1 are gases, the purification of final product is not required.
Lucas Test
The formation of alkyl halides from alcohols is the basis of this test. In involves the reaction of the alcohol with Lucas reagent (anhyd. ZnCl2 + conc. HCl). Since the reactivity of alcohols is in the following order:
primary alcohols < secondary alcohols < tertiary alcohols
With primary alcohols turbidity does not appear. In case of secondary alcohols, turbidity appears within 5 minutes whereas it appears immediately with tertiary alcohols. The turbidity is due to the formation of alkyl chlorides from the corresponding alcohols.