Galvanic Cell
Galvanic cells are called voltaic cells. In such a cell chemical energy is converted into electrical energy. Dry cells, car batteries and button cells used in wrist watches are all examples of this type of cell. They are energy producing devices.
Redox Reaction and Galvanic Cell
When a Zinc rod is dipped in CuSO4 solution, a reaction starts in the solution.
Zn (s) + CuSO4 (aq) → ZnSO4 (aq) + Cu (s)
It is an example of redox reaction. The two half reactions are:
Zn (s) → Zn2+ (aq) + 2e– (Oxidation)
Cu2+ (aq) + 2e– → Cu(s) (Reduction)
In this redox reaction the electrons given by zinc rod have been directly consumed by Cu2+ ion. But, if somehow the electrons given by Zinc rod are made to flow through a wire to reach Cu2+ ions, electric current can be produced. To do so, the reaction is carried out in the electrochemical cell.
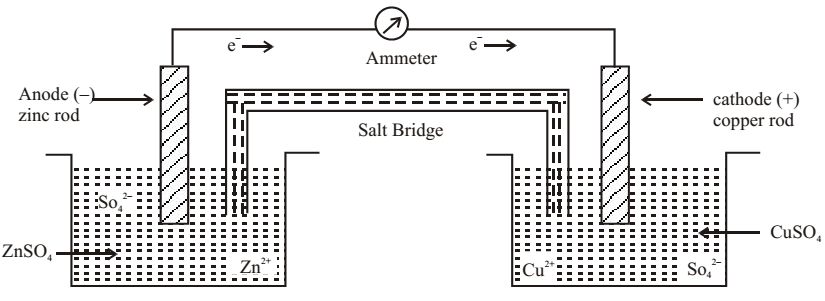
Zinc rod is dipped in zinc sulphate solution in one beaker while copper rod is dipped in another beaker containing CuSO4 solution. The two solutions are connected through a salt bridge and the two metals are connected to an ammeter with the help of wire. Electrons move through the wire from zinc to copper rod.
A metal dipped in its own salt solution is called as half cell. Zinc rod dipped in-zinc sulphate solution is oxidation half cell because oxidation takes place.
Zn (s) → Zn2+ (aq) + 2e– (Oxidation)
The released electrons are taken up by zinc rod and it becomes negatively charged.
Copper in copper sulphate is reduction half cell. Copper acts as cathode and reduction take place here. Copper rod becomes positively charged. Copper gains electrons and in this process, becomes positively charged.
Cu2+ (aq) + 2e– → Cu(s) (Reduction)
The electrons will move from negatively charged electrode to positively charged copper electrode. Since the electric current always move in a closed circuit salt bridge is used to make electrical contact between the two half cells.
Symbolic Representation
The rules of notations are:
1. Anode is written on the left hand side and cathode on the right hand side.
2. The metal electrode in the anode half cell is written by its symbol and this is followed by the cation (metal ion) along with its concentration in a small bracket. The metal and cations are separated by vertical line or a semicolon.
Zn (s) | Zn2+ (aq) (1M)
3. In the reduction half cell the anion along with its concentration is written first, then vertical line and then the metal.
Cu2+ (aq) (1M) | Cu (s)
4. A salt bridge is represented by two vertical lines.
Zn (s) | Zn2+ (aq) (1M) || Cu2+ (aq) (1M) | Cu (s)