Heisenberg’s Uncertainty Principle
An important consequence of the wave-particle duality of matter and radiation was discovered by Werner Heisenberg in 1927 and is called the uncertainty principle. According to this principle, it is not possible to simultaneously measure both the position and momentum (or velocity) of an electron accurately.
Mathematically, the Heisenberg principle can be expressed in terms of an inequality.
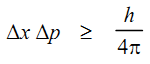
Where Δx and Δp are the uncertainty in the measurements of position and momentum respectively. If the position of an object is known exactly (Δx = 0), then the uncertainty in the momentum must be infinite, meaning that you cannot say anything about the velocity. Similarly, if the velocity is known exactly, then the position would be entirely uncertain and the particle could be anywhere. In actual practice none of the two properties can be measured with certainty.
Due to the small value of the Planck’s constant (h = 6.626 x 10-34 J s) this principle is not relevant while making measurements of large objects like car, bus, etc. It is relevant, only when you are making measurements on very small objects such as electrons.
Heisenberg’s principle questioned the validity of Bohr’s model. It is so because according to Bohr’s model you can precisely calculate the radius of the orbit (the position of the electron) and the velocity of electron in it. But, it is not possible according to Heisenberg’s principle.
It motivated many scientists to develop newer models of the atom using the dual nature of the electron. This resulted into the development of a Quantum mechanical or Wave Mechanical Model of the atom.