Hydrogen Bonding
It is a special type of attraction between a hydrogen atom bonded to a strongly electronegative atom (like nitrogen, oxygen or fluorine) and the unshared pair of electrons on another electronegative atom.
Hydrogen bond is a weak bond, the strength being just about 4-25 kJ mol–1. It is quite small as compared to the covalent bond, which needs a few hundreds of kJ mol–1 of energy to break. However, it is strong enough to be responsible for the high boiling points of H2O and HF.
It is due to hydrogen bonding only that water exists as a liquid. The low density of ice also can be explained in terms of hydrogen bonding.
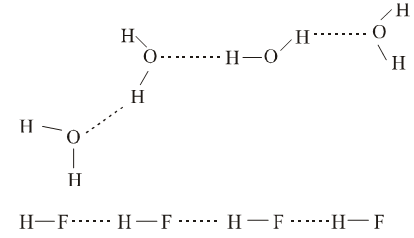
Due to the difference in the electronegativity between hydrogen and the other electronegative atom, the bond connecting them becomes polar. The hydrogen atom acquires a positive charge while the electronegative atom bears the negative charge. Hydrogen bonding results from the electrostatic interaction between the positively charged hydrogen atom and the negatively charged electronegative atom.
The second electronegative atom may be a part of the same molecule or it may belong to a different molecule. Accordingly, there are two types of hydrogen bonds:
- Inter-molecular Hydrogen Bonding
- Intra-molecular Hydrogen Bonding
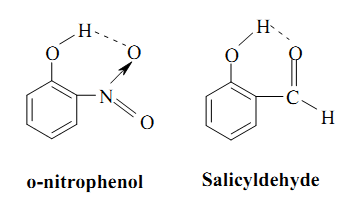
Salicyldehyde and o-nitrophenol are two common examples of the molecules showing intramolecular hydrogen bonding whereas in water, intermolecular hydrogen bonding exists.