Ionization of Weak Bases
The ionization of weak bases (BOH) can be expressed as:
BOH (aq) ⇌ B+ (aq) + OH- (aq)
The equilibrium constant expression for the reaction is
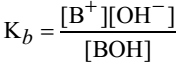
The constant Kb is called dissociation constant of the base. It gives the idea about the relative strengths of weak bases. Higher the value of Kb the stronger is the base.