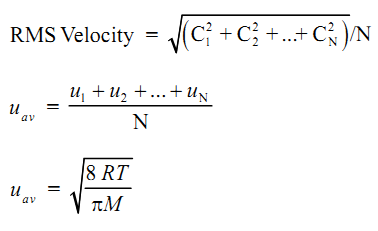Kinetic Molecular Theory of Gases
To explain the behavior of the gases theoretically, Claussius, Maxwell and Boltzmann made the following assumptions:
- Gases consist of large number of tiny particles called molecules.
- The gas molecules are so small and so far apart that the total volume of the molecules is a negligible fraction of the total volume occupied by the gas.
- The molecules are in a state of constant, rapid and random motion colliding with one another and with the walls of the container.
- There are no attractive or repulsive forces between the molecules of the gas.
- The collisions of the molecules among themselves and with the walls of the containing vessel are perfectly elastic, so that there is no loss of energy during collisions.
- The pressure exerted by a gas is due to the bombardment of the molecules on the walls of the containing vessel.
- The kinetic energy of a gas is a directly proportional to the absolute temperature of the gas.
On the basis of this model, it is possible to derive the following expression for a gas:
pV = 1/3 × mNC2
Where p is pressure, V denotes volume, m is the mass of a gas molecule, N is the total number of molecules, and C is the root mean square velocity of the gas molecules.
Root Mean Square and Average Velocity
Root mean square velocity is the square root of the average of the squares of all the molecular velocities.