Oxidation of Alkenes with Ozone
Ozone adds to the alkene forming ozonide. The ozonide when further reacted with water in the presence of zinc dust, forms aldehydes or ketones, or both.

The process of addition of ozone to an unsaturated hydrocarbon followed by hydrolysis is called ozonolysis. Ozonolysis can be used for the determination of the position of double bonds in alkenes by analysing the products formed (aldehydes and ketones).
When but-1-ene is oxidized with ozone and the ozonide formed is hydrolysed, we get one mole of propanal and one mole of methanal, showing that the double bond is between carbon atom 1 and 2.

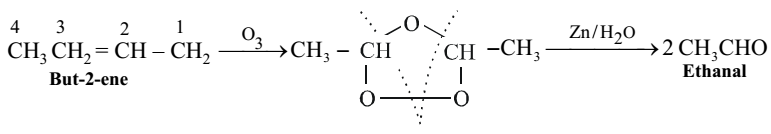
When but-2-ene on oxidation with ozone, followed by hydrolysis, gives two moles of ethanal, showing that the double bond is present between carbon atoms 2 and 3.