Raoult’s Law for Solutions
Raoult’s law states that for a solution of volatile liquids, the partial vapour pressure of each liquid in the solution is directly proportional to its mole fraction. Raoult’s law is applicable only if the liquids are miscible (homogenous mixture).
The vapour phase now consists of vapours of both the liquids A and B. The partial vapour pressure of each liquid will depend upon its mole fraction in the solution. Let the mole fractions of the liquids A and B be XA and XB respectively. Also, if PA and PB are the partial vapour pressures of A and B respectively, then
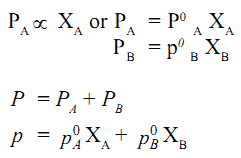
A solution which obeys Raoult’s law over the entire range of concentration at all temperatures is called an ideal solution.