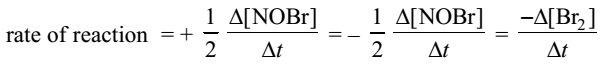Rate of a Chemical Reaction
When the reactants are mixed to perform a reaction, there are no products present initially. As time passes, the concentration of the products increases and that of the reactants decreases. The rate of any chemical reaction can be expressed as the rate of the change in concentration of a reactant (or a product).
In order to make the rates with respect to different reactants or products equal, the rate expression is divided by the stoichiometric coefficient in the balanced chemical equation.
For example, in the equation,
2NO (g) + Br2 (g) → 2NOBr (g)
The rate of reaction with respect to reactants and products is expressed as: