Reduction of Carbonyl Compounds At α-Carbon
The α-hydrogen in aldehydes and ketones is quite acidic and can be easily abstrated by a strong base. The resulting anion can stabilize by resonance. It is called an enolate ion. On protonation, it gives an enol. Thus, keto form and enol form are in equilibrium. This is also known as keto-enol tautomerism.
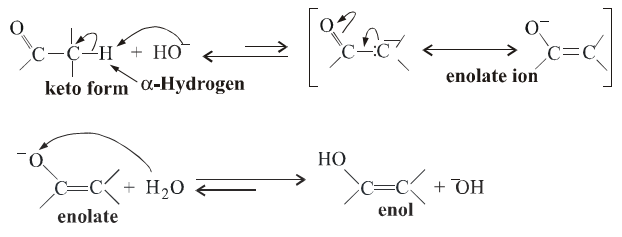
Other reactions feasible due to the presence of α-hydrogen are Halogenation and Aldol Condensation.