Rutherford’s Gold Foil Experiment
Ernest Rutherford performed an experiment called Gold Foil Experiment or α-ray scattering experiment to test the structure of an atom as proposed by Thomson.
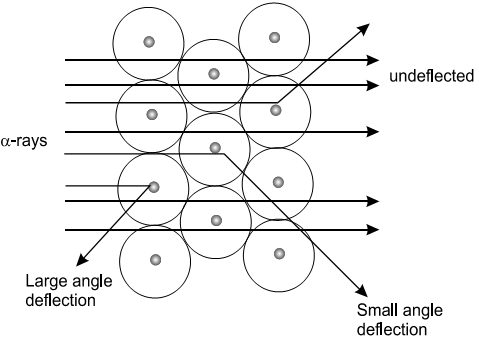
In this experiment, a beam of fast moving alpha particles (positively charged helium ions) was passed through a very thin foil of gold. He expected that the alpha particles would just pass straight through the gold foil and could be detected by a photographic plate. But, the actual results of the experiment were quite surprising.

It was observed that most of the α-particles did pass straight through the foil but a number of particles were deflected from their path. Some of these deflected slightly while a few deflected through large angles and about 1 in 10,000 α-particles suffered a rebound.
These results led Rutherford to conclude that:
- The atom contained some dense and positively charged region located at the center of the atom that he called as nucleus.
- All the positive charge of the atom and most of its mass was contained in the nucleus.
- The rest of the atom must be empty space which contains the much smaller and negatively charged electrons.
However, there was a problem with the Rutherford’s model. According to the Maxwell’s theory of electromagnetic radiation, a charged particle undergoing acceleration would continuously emit radiation and lose energy. Since the electron in the atom is also a charged particle and is under acceleration, it is expected to continuously lose energy. As a consequence, the electron moving around the nucleus would approach the nucleus by a spiral path and the atom would collapse. However, since it does not happen, the Rutherford’s model failed to explain the stability of the atom.
The next attempt to suggest a model for atom was made by Neils Bohr, a student of Rutherford. This model used the concept of quantization of energy of electrons in the atom.