Separation of Coloured Components by Paper Chromatography
Chromatography is a technique by which a mixture of various substances can be separated, purified and identified. The term chromatography is applied to separation processes based on the principle of distribution of a sample between two phases:
- a stationary phase or a fixed phase
- a moving phase
There are two variations in the types:
- solid-liquid chromatography
- liquid-liquid chromatography
In solid-liquid chromatography, the solid surface adsorbs the substance which is loaded (charged) on the surface and the compounds are separated on the solid surface. It can also be classified as
- (i) adsorption chromatography
- (ii) partition chromatography
- (iii) ion-exchange chromatography
Adsorption chromatography is based on selective adsorption of substances on the surface of an adsorbent. Column chromatography, thin layer chromatography, etc. are based on the adsorption phenomenon. In column chromatography, the stationary phase is an adsorbent and the mobile phase is a solvent.
Liquid-liquid chromatography involves the distribution of a substance between two immiscible solvents to different extents. Paper chromatography is an example of liquid-liquid chromatography. The stationary phase is supported by sheet of filter paper. The filter paper, known as chromatography paper, holds water which acts as a stationary phase. The mobile phase is another liquid which carries the substance on the paper along with it.
Every substance has a characteristic Rf value at a given temperature and for a given solvent.
Rf stands for Ratio of Fronts or the retention factor. It is the ratio of the distance traveled by the component to the distance traveled by the solvent, from the origin.

Experiment
Separation of the coloured components of the following by paper chromatography and comparison of their Rf values.
- (a) Black ink or a mixture of red and blue inks
- (b) Juice of a flower or grass
How To Perform Experiment
To separate the components of a mixture of red and blue inks and find out their Rf values.
Take the Whatmann filter paper strip (25 × 2 cm). Draw a line with a pencil about 4 cm above the lower end.
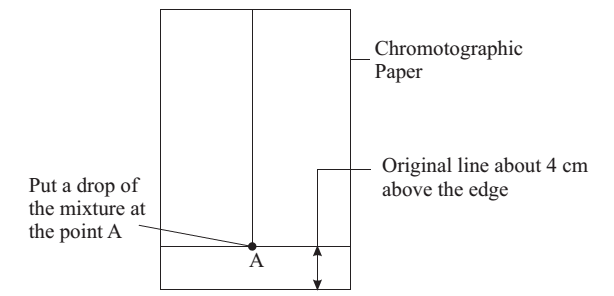
Spotting: With the help of a fine capillary tube, put a drop of the mixture of red and blue inks at point A on the filter paper strip. Let the spot dry in air.
Put another drop on the same spot and dry again. Repeat the process 2-3 times to make the spot rich in the mixture. Suspend the filter paper vertically in the gas jar containing the solvent mixture (50% alcohol). Hang the paper from the glass rod with the help of clips or hook in such a way that the spot remains about 2 cm above the solvent level. Alternatively cork with hook may be used.
Cover the jar with a glass cover and keep it undisturbed. Notice the rise of the solvent as it ascends the paper. After the solvent has risen about 20 cm, you will see two different spots of red and blue colours on the filter paper. Take the filter paper out of the jar and mark the level of the solvent with a pencil. Dry the paper.
Mark the centres of the blue and the red spots. Measure the distance of the two spots and that of the solvent from the initial line. Record your observations and calculate the Rf values of the red and blue spots.
Precautions
- Use very fine capillary tube for spotting. Otherwise the spot will become too big.
- Allow the spot to dry before putting another drop. A good spot is small and does not contain too much of the mixture.
- Do not press the capillary tube very hard, otherwise it may pierce the paper or break. If you make a hole in the paper then the compound will not rise.
- Do not allow the paper to curl at the ends. It should hang straight, without touching the sides of the jar.
- Keep the jar covered. Do not disturb the jar after hanging the filter paper till the solvent has risen up to the desired level.
- The spot should not dip in the solvent. Otherwise the compounds will dissolve in it.
- Use distilled water for making the solvent mixture to be taken in the jar.