Structures of Ionic Solids
In case of ionic solids that consist of ions of different sizes, you need to specify the positions of both the cations as well as the anions in the crystal lattice. Therefore, structure adopted by an ionic solid depends on the relative sizes of the two ions. It depends on the ratios of their radii (r+/r-) called radius ratio.
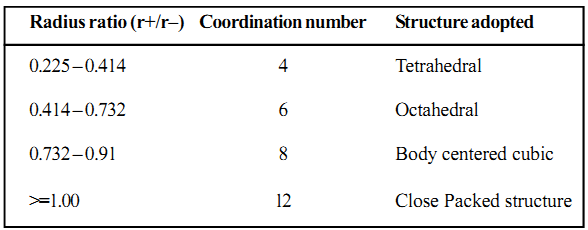
The common ionic compounds have the general formula as MX, MX2, and MX3, where M represents the metal ion and X denotes the anion.