Vapor Pressure and Boiling Point
The extent of evaporation of a liquid is measured with the help of vapor pressure of a liquid.
Vapor Pressure
A liquid placed in an open vessel evaporates completely. However, if the liquid is allowed to evaporate in a closed vessel, evaporation occurs, but after sometime the level of the liquid does not change any further and become constant.
In the closed vessel, the molecules evaporating from the liquid surface are confined to a limited space. These molecules may collide among themselves or with the molecules of air and some of them may start moving towards the surface of the liquid and enter into it. This is known as condensation.
In the beginning, rate of evaporation is greater than the rate of condensation. But as more and more molecules accumulate in the space above the liquid, rate of condensation gradually increases. After some time, rate of evaporation becomes equal to the rate of condensation and an equilibrium state is reached. The number of molecules in the vapor above the liquid becomes constant.
These molecules exert certain pressure over the surface of the liquid. This pressure is known as vapor pressure. The vapor pressure of a liquid has a characteristic value at a given temperature. For example, vapor pressure of water is 17.5 torr and that of benzene is 75.00 torr at 20º C.
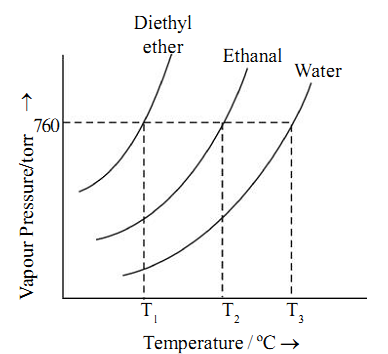
The vapor pressure of a liquid increases with increase in temperature. It is so because at a higher temperature more molecules have sufficiently high energy to overcome the forces of attraction and escape to form vapor. A plot of vapor pressure as a function of temperature is called vapor pressure curve.
Boiling
The temperature at which boiling occurs is called the boiling point of the liquid. At this temperature the vapor pressure of the liquid is equal to the atmospheric pressure. The boiling point depends upon the atmospheric pressure. For example, water boils at 100ºC at 760 torr and at 97.7ºC at 700 torr.
The normal boiling point of a liquid is defined as the temperature at which the vapor pressure of a liquid is equal to one atmosphere or 760 torr.
The boiling point of a liquid depends upon its nature. A more volatile liquid would boil at a lower temperature than a less volatile liquid.
Evaporation and Boiling
Evaporation and boiling, both involve conversion of a liquid into vapor and appear to be similar. However, they differ from each other in some aspects. Evaporation occurs at all temperatures from freezing point of a liquid to its boiling point, while boiling occurs at a definite temperature only (boiling point). Evaporation occurs slowly while boiling is a fast process. Evaporation of a liquid occurs at its surface alone while boiling occurs throughout the liquid.