Variation of Cell Potential With Change in Concentration of Electrolytes
The EMF of a cell varies with the concentration of the two electrolyte solutions according to the following Nernst equation:
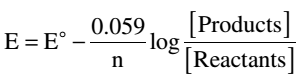
where E = the electromotive force of the cell E° = the standard electromotive force of the cell
In the case of the Daniel cell:

Experiment: To study the variation of cell potential in Zn | Zn2+ || Cu2+ | Cu with change in concentration of electrolytes (CuSO4 or ZnSO4) at room temperature.
Apparatus Required
100 ml beakers, 250 ml beaker, measuring flask, 100 ml measuring cylinder, connecting lead, voltmeter, U tubes, cotton.
Chemicals Required
Copper strip, zinc strip, copper sulphate, zinc sulphate, potassium chloride.
How To Perform Experiment
(a) Preparation of solutions copper sulphate
1. Weigh 29.969 g of copper sulphate on a watch glass. Transfer it to a 250 ml beaker and dissolve it in water (sufficiently less than 100 ml). Transfer the solution into 100 ml measuring flask and make its volume to 100 ml. It is 1.00 M CuSO4 solution and store it. Mark it A.
2. Prepare 1.00 ml copper sulphate solution each of 0.5 M, 0.25 M, 0.125 M and 0.0625 M strength by dilution as follows.
To prepare 0.5 M CuSO4 solution, take 50 ml of 1.0 M CuSO4 solution prepared in step 1 in another measuring flask with the help of measuring cylinder make its volume to 1.00 ml. Mark it as B.
Similarly, you can prepare the solution of 0.25 M, 0.125 M and 0.0625 M.
(b) Preparation of zinc sulphate solution
Prepare 100 ml 1.0 M zinc sulphate solution by dissolving 28.756 g zinc sulphate in water.
(c) Preparation of salt bridges
(i) Take a U tube of glass of about 10 cm length.
(ii) Dissolve about 2.0 g of agar agar completely in about 25 ml of water taken in a borosil beaker by heating it at a low flame.
(iii) Dissolve about 12 g of solid potassium chloride (KCl) completely in the agar agar solution in water by stirring.
(iv) Fill the U tube with it and allow it to remain in an upright position till agar agar gets set.
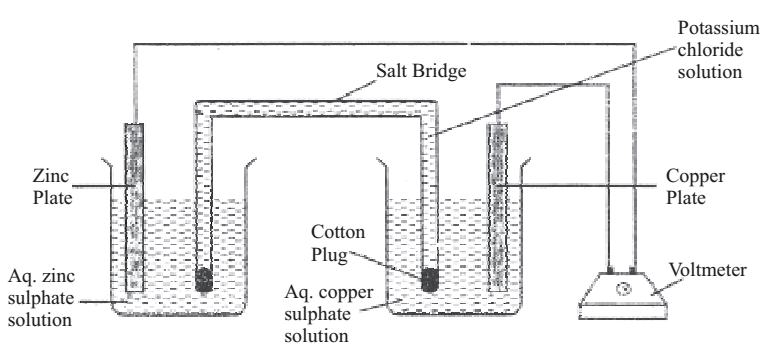
(d) Setting up of simple electrochemical cells and measuring its EMF
Assemble the electrochemical cells with different combinations of concentrations.
Zn (s) | Zn2+ (aq) || Cu2+ (aq) | Cu (s)
Use separate salt bridges for different readings.
(i) Take 30 ml of 1.0 M copper sulphate solution (from flask A) in a 100 ml beaker and 30 ml of 1.0 M zinc sulphate solution in another 100 ml beaker.
(ii) Immerse the metal strips in solutions containing the same metal ions.
(iii) Connect both the metal strips to each other through a voltmeter with the help of connecting lead.
(iv) Connect both the solutions through salt bridge.
(v) Note the voltmeter reading that gives the E.M.F of the cell.
(vi) Repeat the same procedure with copper sulphate solutions of different concentrations using 0.1 M solution of zinc sulphate.
Precautions
- (i) Electrodes should be cleaned by sand paper.
- (ii) There should be no air bubble in the salt bridge.
- (iii) Use separate salt bridge for every cell.
- (iv) Each voltmeter reading should be taken after stirring the solution.