Viscosity
Every liquid has the ability to flow. It is due to the fact that molecules in a liquid move freely, although within a limited space. Water flows down a hill under gravitational force or through pipes when forced by a pump. Some external force is always required for a liquid to flow.
Some liquids like glycerol or honey flow slowly while others like water and alcohol flow rapidly. This difference is due to the internal resistance to flow which is called viscosity.
The liquids with higher viscosity flow slowly and are more viscous in nature like glycerol or honey. Water and alcohol have lower viscosity and are less viscous in nature. They flow more rapidly.
The viscosity is related to the inter-molecular forces. Stronger the inter-molecular forces more viscous are the liquids. When a liquid flows steadily, it flows in different layers with one layer sliding over the other. Such a flow is known as laminar flow.
As the distance of the layer from the surface increases, velocity increases. Thus, different layers move with different velocities. Due to inter-molecular forces (cohesive forces) each layer experiences a force of friction from its adjacent layers. This force of friction, f between two layers depends upon:
- area of contact between them, A
- distance between the layers, dx
- difference in velocity between the layers, du
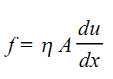
Coefficient of viscosity (η) is the force of friction between two parallel layer of the liquid which have 1 cm2 area of contact, are separated by 1 cm and have a velocity difference of 1 cm s–1.
Units of Viscosity
CGS unit of viscosity is dyne cm–2 s. This unit is also known as poise (P). The SI unit of viscosity is N m–2 s or Pa s.
Effect of Temperature
Viscosity of a liquid decreases on raising the temperature. It is due to decrease in inter-molecular forces on heating.